QC Laboratories
LIMS – End-to-End Automation of your
Laboratory Processes for greater Efficiency
and Product Quality
As experienced implementation specialists, we understand the challenges that
quality control (QC) laboratories face today. up2LIMS is a catalyst for process optimization specifically designed to comprehensively support and meet these requirements in quality control.
Faster
Product Development Cycles
The implementation of a LIMS enables efficient and meaningful tests that promote faster and safer product development cycles. With integrated analysis tools and flexible workflows, up2LIMS ensures that all QC-relevant data can be recorded and evaluated. Automated reporting and real-time data access enable companies to react quickly to changes and make informed decisions.
Compliance with
Regulatory Requirements
up2LIMS is designed to meet regulatory requirements. Our solution supports compliance with GLP, GMP and other relevant standards, for example through seamless logging and traceability of all laboratory activities.
The LIMS offers comprehensive audit trails and role-based access controls to ensure the integrity of the data in the long term.
Intelligent
Networking of Business Units
By combining development, production and quality control data, up2LIMS provides a central platform for data management. The LIMS software thus enables seamless integration of data across the entire product life cycle. This not only optimizes collaboration between different departments, but also improves the transparency and traceability of internal processes.
Automation
and optimal Use of Resources
In order to withstand cost pressure, up2LIMS relies on comprehensive automation solutions. The application supports automation of routine tasks and processes, which leads to an increase in efficiency and at the same time minimizes human error. Transparent evaluation mechanisms support the optimal use of all laboratory resources, which contributes to a general reduction in costs.
Precise Planning &
Continuous Optimization
With up2LIMS, QC laboratories can plan their processes precisely and continuously expand and optimize them. The solution offers tools for process analysis and monitoring to identify bottlenecks and recognize potential for improvement, including in cross-departmental collaboration. Regular evaluations and internal reports enable laboratories to continuously improve their processes and adapt them to changing requirements.
Optimal Balance of
Quality, Safety and Efficiency
The LIMS application is designed to maintain an optimal balance between quality, safety, and efficiency. up2LIMS ensures that all processes meet the highest quality and safety standards while increasing laboratory efficiency. The seamless integration of QC laboratory and production through up2LIMS offers companies decisive competitive advantages by optimizing data flow, traceability and decision making.
up2LIMS at a glance:

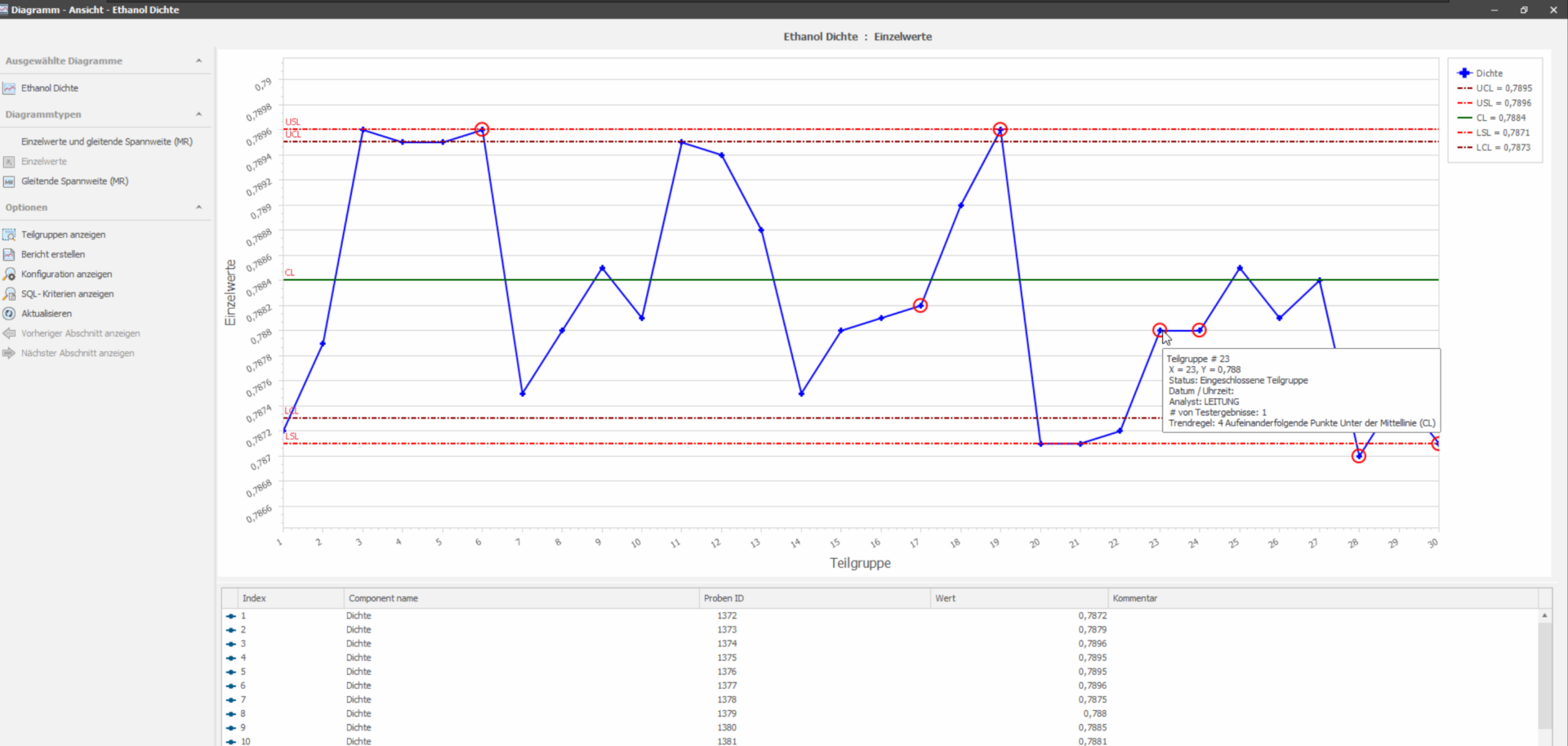
Statistical Quality Control Module
The “SQC” (Statistical Quality Control) add-on module offers comprehensive options for monitoring and analyzing processes in QC laboratories to ensure continuous monitoring of product quality. It primarily enables the calculation and graphical display of process or product parameters. The configurable selection criteria for selecting the result values allow a high degree of flexibility when defining the respective charts. This enables versatile evaluations of product-related parameters from production as well as values for control samples, including control charts, process capability and trend analysis. The module offers standard-compliant monitoring of product quality and ensures transparency and automation throughout the entire product life cycle thanks to seamless integration into production recording systems.

Your LIMS project starts here!
Get to know up2LIMS: The All-In-One LIMS that fits today and grows with your requirements tomorrow – fair, flexible and GxP-ready.
Your LIMS project starts here!
Get to know up2LIMS: The All-In-One LIMS that fits today and grows with your requirements tomorrow – fair, flexible and GxP-ready.