Pharmaceutical Laboratories
LIMS – Ensures Quality, Compliance,
and Increased Efficiency
We have been a reliable partner for pharmaceutical companies of all sizes for decades. With numerous successful projects in the fields of pharmaceuticals, medical devices and active ingredients, we have established ourselves in this industry. Our LIMS solution is specially designed to optimally support the special requirements of pharmaceutical laboratories.
Compliance
with the GxP-Guidelines
Pharmaceutical laboratories must adhere to strict GxP-guidelines, including Good Laboratory Practice (GLP), Good Manufacturing Practice (GMP) and Good Clinical Practice (GCP). Our LIMS ensures compliance with these guidelines through comprehensive documentation and tracking capabilities. We offer integrated audit trails, role-based access controls and automated logging to ensure that all data is captured completely and accurately.
Ensuring
Data Integrity
Data integrity is critical throughout the lifecycle of a drug – from development to manufacturing to clinical trials. Our LIMS solution provides secure and integrable systems for data collection, storage, and analysis. By implementing validation processes and regular compliance testing, we ensure that all data is reliable, consistent, and accurate.
Validation & Calibration
of Instruments and Devices
Our LIMS solution supports the proper validation and calibration of laboratory instruments and equipment. Through automated reminders and logging of calibration and maintenance activities, we ensure that all devices are always accurate and ready for use. This contributes significantly to the accuracy of test results and minimizes the risk of measurement errors.
Implementation of strict
Quality Assurance Measures
Strict quality assurance measures are essential to ensure the integrity of pharmaceutical laboratory data. Our LIMS solution offers comprehensive quality assurance capabilities, including quality document management, deviation management and CAPA (Corrective and Preventive Actions) management. This enables pharmaceutical laboratories to identify potential risks at an early stage and take proactive measures to prevent errors.
Flexible & Scalable
for innovative Requirements
Pharmaceutical laboratories must continuously adapt to meet the highest standards. Our LIMS solution is flexible and scalable, allowing you to adapt to changing requirements and technological advances. Through regular updates and improvements, we ensure that our solution is always up to date and meets the current requirements of the industry.
Audit-proof centralized
Document Management
As a central platform, the LIMS takes over the audit-proof management of all laboratory-specific documents such as SOPs, test protocols and certificates of analysis. Through version control and regulated access rights, the system ensures that changes are made in a traceable and controlled manner. The linking of analysis data with associated documents ensures complete traceability and simplifies audits. Document processes are automated and sources of error are reduced so that GxP requirements can be met efficiently.
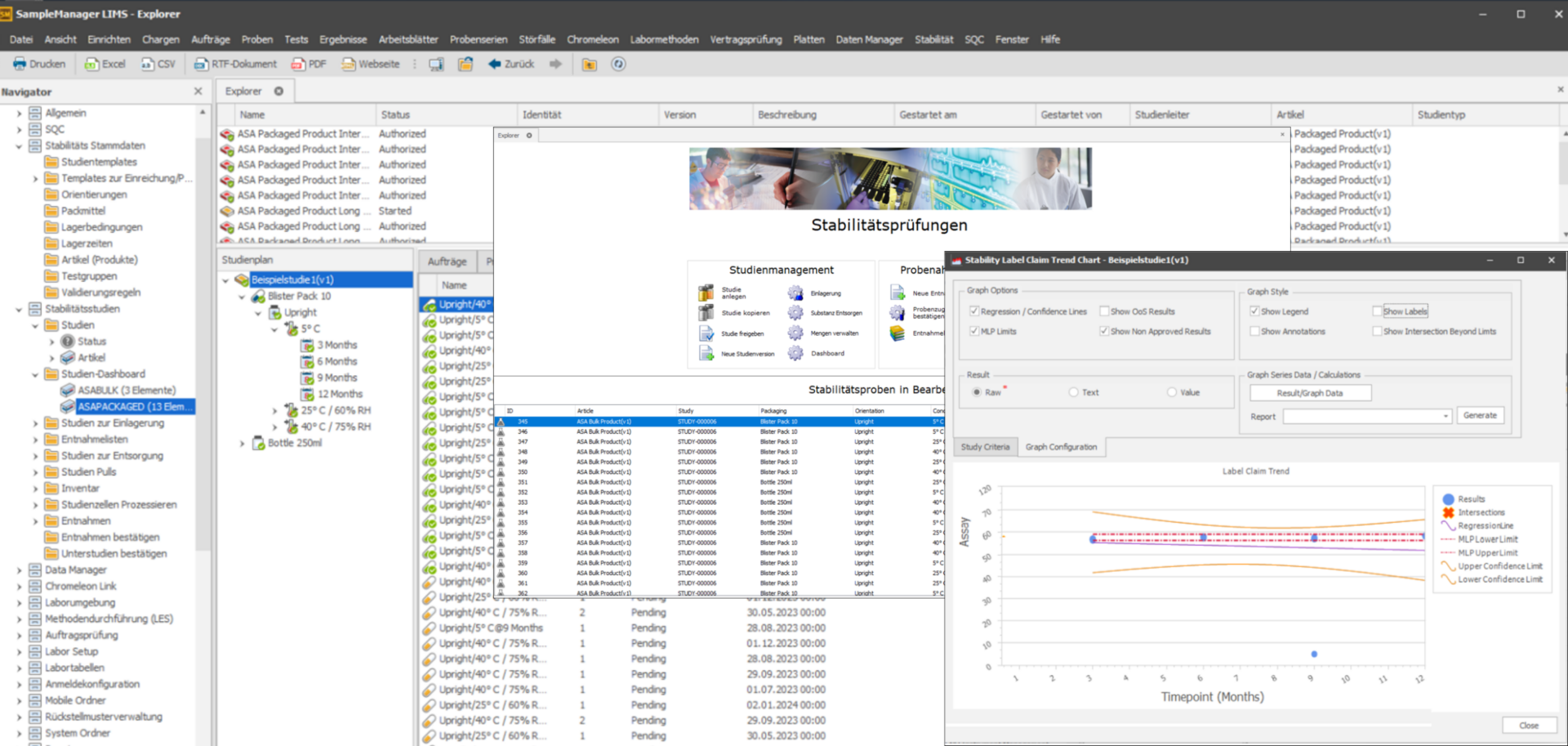
LIMS Stability Module
Stability studies are critical to ensure the quality, safety, and efficacy of medicinal products throughout their shelf life. These studies identify potential degradation mechanisms under different environmental conditions and allow manufacturers to set appropriate expiry dates. The stability management module enables the administration of stability studies from study design to study completion.
The system supports study planning and design, enabling GxP-compliant adjustments with full traceability of all changes made. It precisely calculates the required sample quantity and manages the stored quantities; all withdrawals are fully documented. Dashboards enable simple status tracking in real time. Flexible workflows support adaptation to a wide range of laboratory requirements. A high degree of configurability ensures optimum adaptation to the respective priorities of pharmaceutical laboratories.

Your LIMS project starts here!
Get to know up2LIMS: The All-In-One LIMS that fits today and grows with your requirements tomorrow – fair, flexible and GxP-ready.
Your LIMS project starts here!
Get to know up2LIMS: The All-In-One LIMS that fits today and grows with your requirements tomorrow – fair, flexible and GxP-ready.