up2LIMS
All-in-One LIMS for Small Lab Units
up2LIMS – One Software. All Features.
Perfect fit for efficient laboratory work.
Simple and precise to use, scalable to your requirements and successfully implemented in laboratories of all sizes and in all sectors.
- Flexible and customized, even for small and medium-sized laboratories
- Complete process coverage
- Compliant with the ISO 9001:2015 standard and other legal and regulatory guidelines
- Fast implementation, ready to use immediately
- Fair, transparent pricing
- Personal support from a single source
- Integrated Laboratory Information Management System (LIMS), Electronic Lab Notebook (ELN), and Lab Execution System (LES) are included
Developed by experienced laboratory experts.
With over 30 years of laboratory expertise and based on the proven technology of Thermo Scientific™ SampleManager LIMS™, up2LIMS offers you future-proof laboratory solutions with in-depth industry know-how.
up2LIMS – at a Glance
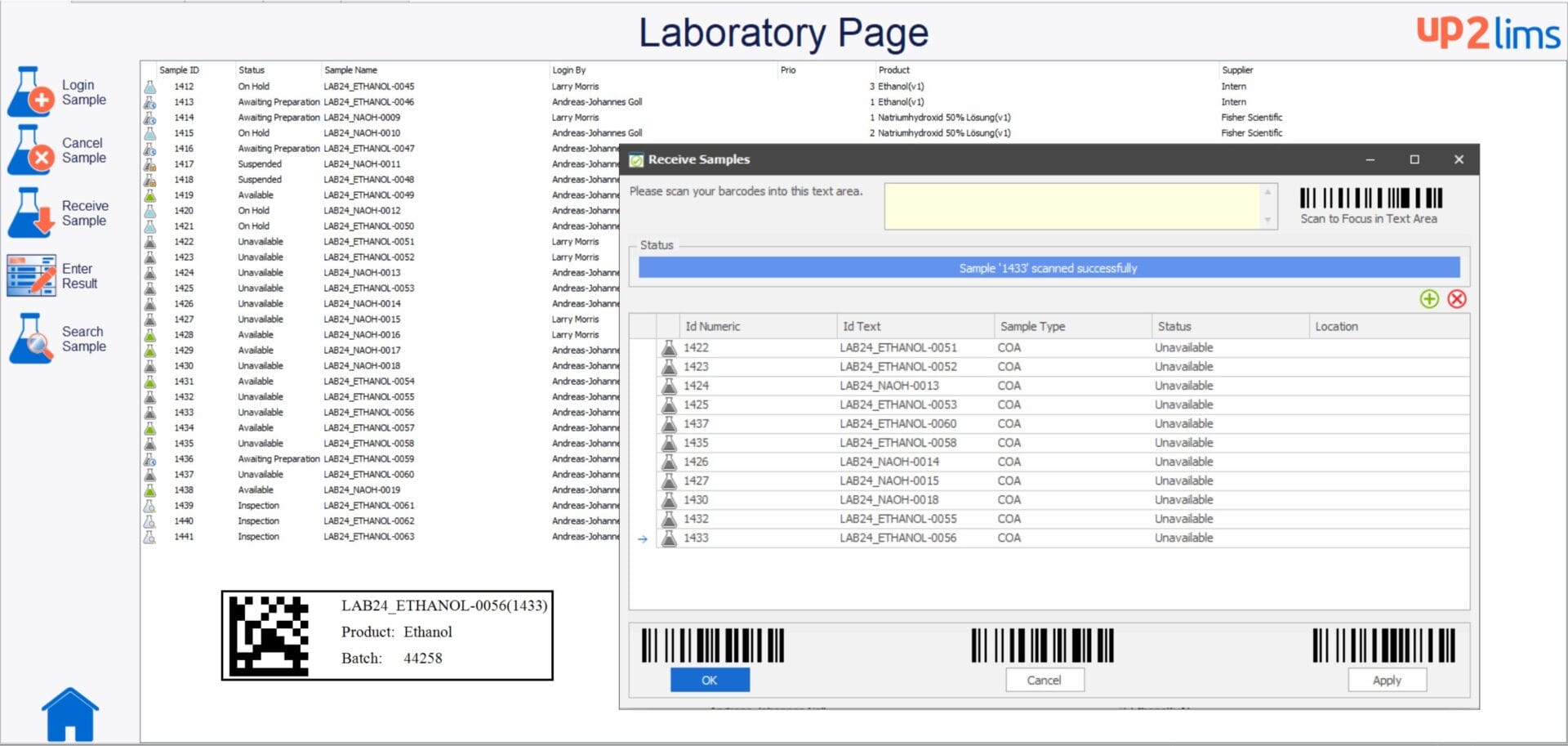
The individually configurable user interfaces enable fast and intuitive operation. up2LIMS supports the flexible recording of inspection orders, and each order type can be configured individually. Once the order has been entered, the scope of testing can be stored directly. Integrated barcode functions enable transparent sample allocation and seamless traceability. The measurement is supported by integrated document management and the Lab Execution System (LES); work instructions are available for viewing at the appropriate point. Results can be recorded manually or via direct device interfaces, including functions such as limit value monitoring and change history. A release process enables multi-level releases and an audit level. Once approved, certificates of analysis and standard reports can be generated automatically.
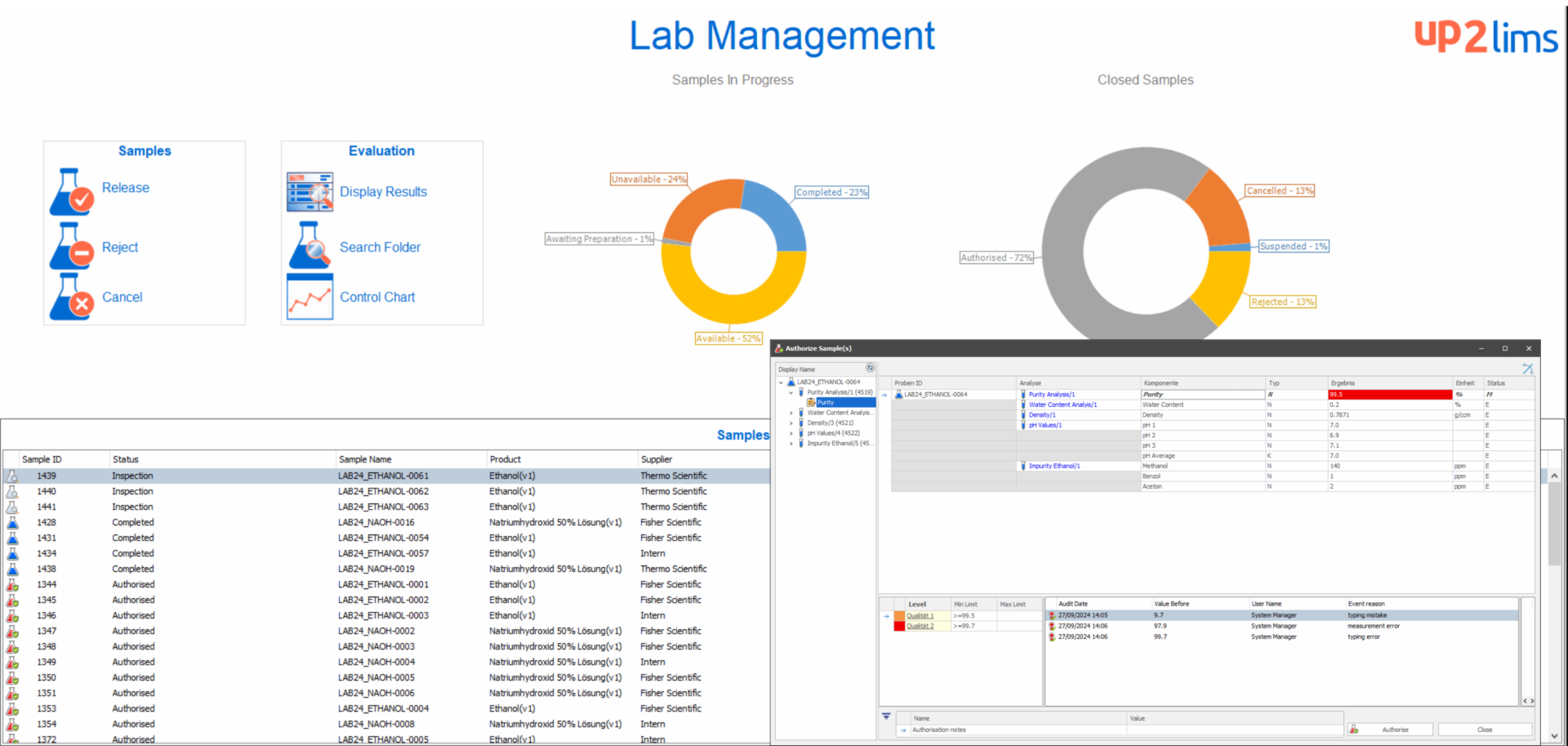
Transparent laboratory management
As a laboratory manager, up2LIMS offers you customized functions for efficient laboratory management. With the help of configurable workflows, you always have an overview of the respective analysis status and your processes. Individual workflows are optimally mapped to ensure maximum transparency, compliance and data integrity. The “SQC module” (Statistical Quality Control) enables the precise calculation and graphical display of process and product parameters, including limit value display and trend analysis. In addition, laboratory performance indicators such as throughput times and error rates are available to you at all times, enabling real-time evaluation and data-supported assessment of the efficiency and quality of your laboratory.
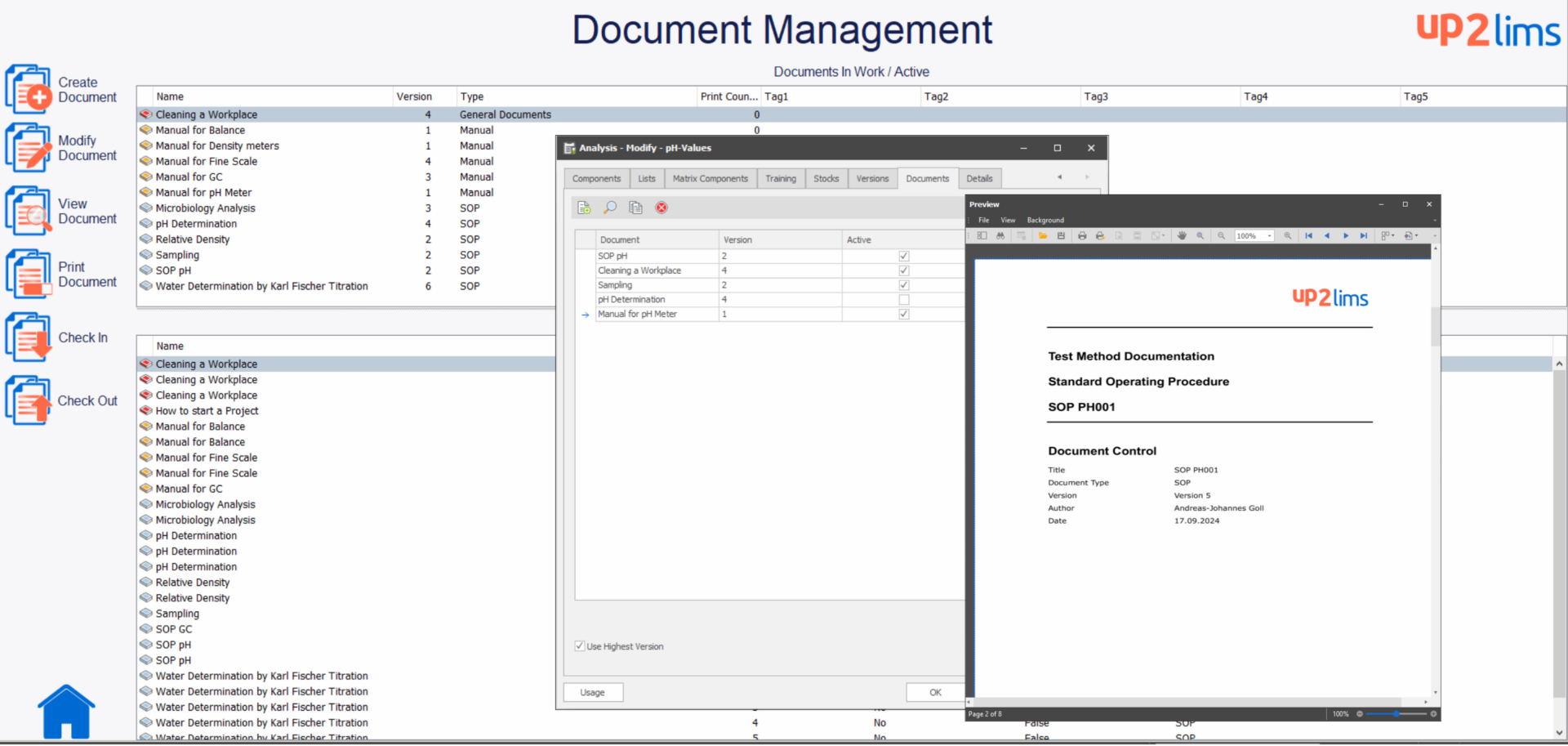
Integrated Document Management
Document management in up2LIMS enables versioned and controlled management of all relevant documents such as work instructions, SOPs and standards. Documents can be referenced at the appropriate place in the LIMS. For example, an SOP can be assigned to an analysis method, a user manual or an instrument. Established document management functions such as keyword search, controlled document processing (versioning, review and release, audit trail) and electronic signatures are already integrated.
Perfect Laboratory Administration
up2LIMS enables the precise recording and management of instruments, reagents, standards, and consumables. This means you always have a complete overview of the condition and availability of your instruments as well as the quantity and expiration date of your reagents. Instrument parts, such as electrodes of a pH meter, can be logged and managed, including the complete maintenance of an instrument logbook. The planning and execution of maintenance and service activities are fully mapped in the LIMS.
up2LIMS has an inventory management system in which all substances and stocks can be transparently mapped from ordering to consumption logging. If stocks fall below a defined level, information can be sent automatically for repeat orders. Hazard warnings in accordance with various standards such as R/S, GHS and NFP 704 can be stored for substances, displayed or printed on labels.
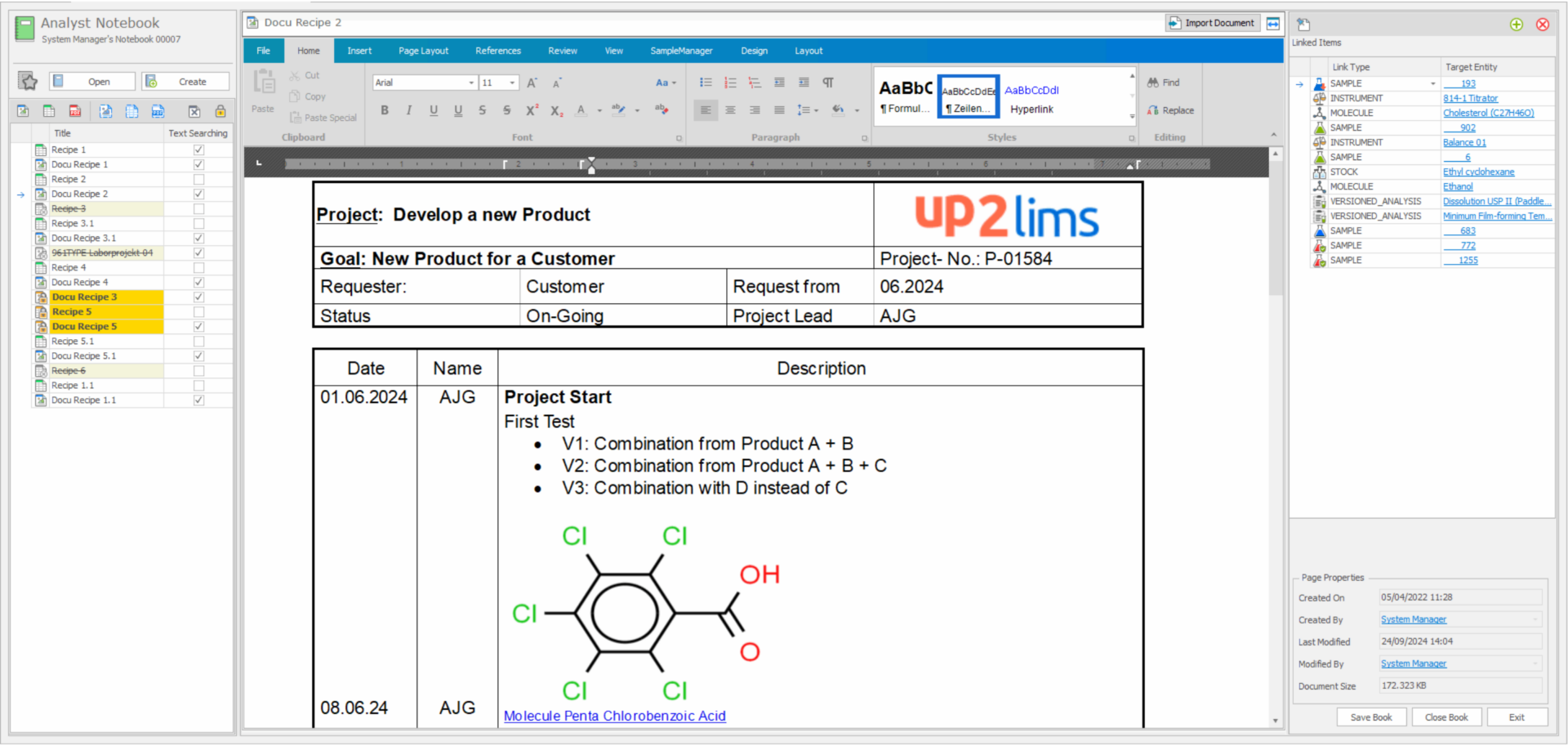
Electronic Lab Notebook
The Lab Notebook (ELN) already integrated in up2LIMS enables you to record unstructured data easily and securely. This data can be referenced in the LIMS so that a direct link to devices and samples can be established at any time. Your Lab Notebook can be set up flexibly according to user or project specifications in a wide variety of formats, images can be inserted or molecular structures referenced. Integrated spreadsheet pages enable calculations and the creation of graphics. All recorded data is available for search functions.
Lab Execution System
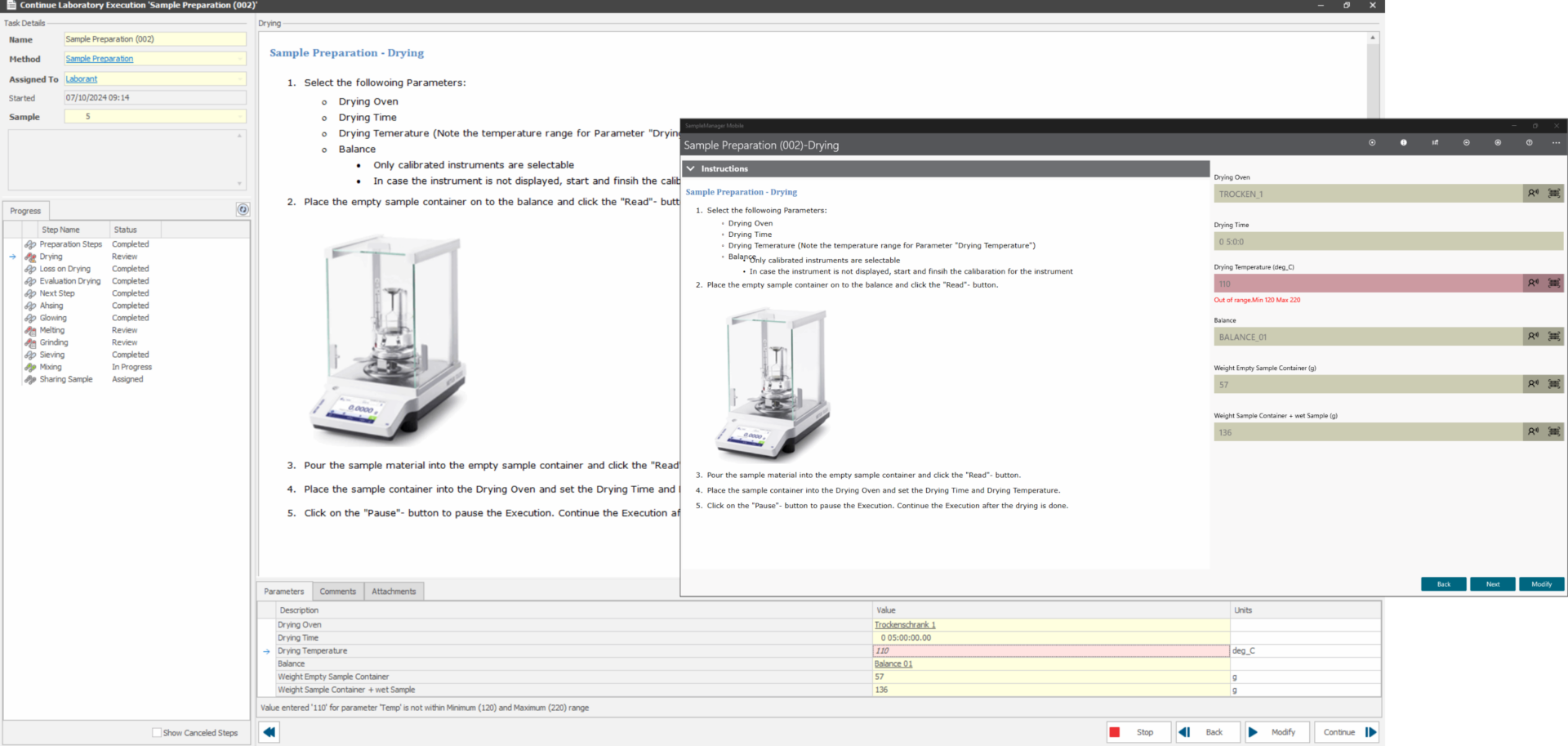
An integrated Lab Execution System (LES) in up2LIMS enables precise and step-by-step execution of various methods such as analyses, instrument calibration or the preparation of reagents. It guides you safely through each required work step, provides the necessary instructions and documents the recorded parameters. With the tablet app, you have maximum freedom of movement in the laboratory and can document the procedure directly at the laboratory workstation.
Seamless Integration of Systems
and Laboratory Equipment
With up2LIMS, your laboratory achieves the maximum of automation, as seamless integration into your systems is guaranteed. In addition, various interfaces to laboratory devices are already included. up2LIMS supports bidirectional interfaces, i.e. the transfer of sample lists or sequences to the device incl. direct retransmission of the results. Of course, the received data can also be processed graphically. You are also provided with options for communicating with other IT systems. File-based communication enables, for example, time- or event-controlled export and import of data. In addition, you are provided with a wide range of options for communicating with other IT systems. For example, file-based communication enables time- or event-controlled export and import of data. Direct read or write access to databases and communication via web services are of course also possible.
Take your Laboratory
to a whole New Level
Innovative automation, end-to-end digitalization, future-proof and highly networked.

Your Local Partner for Laboratory Digitization

The requirements for laboratory processes differ depending on the focus, industry and size. Therefore, no two laboratory workflows are the same. Pharmaceutical laboratories and laboratories in the GxP environment are also confronted with high regulatory requirements. In addition, the specific framework conditions for integrating a LIMS into the existing IT system and device infrastructure must always be taken into account.
We therefore analyze your business processes and specific requirements in order to select the LIMS modules that optimally and efficiently map your laboratory workflow. As a result, we develop exactly the solutions you need to streamline your processes, save costs and ensure compliant work.

Your LIMS project starts here!
Get to know up2LIMS: The All-In-One LIMS that fits today and grows with your requirements tomorrow – fair, flexible and GxP-ready.
Your LIMS project starts here!
Get to know up2LIMS: The All-In-One LIMS that fits today and grows with your requirements tomorrow – fair, flexible and GxP-ready.